Genes are the codes or instructions that tell the body how to grow and develop. Genes are made up of a chemical called DNA and are arranged on strings of information called chromosomes. We all have two copies of each chromosome, one from the egg and one from the sperm that created us. As a result, we have two copies of most genes.
Autosomal Recessive genetic diseases are those that result when both copies of a gene a person has inherited has been altered or changed in a way to make the genes non-working. Everyone carries a few of these altered genes.
A child who inherits one altered or non-working gene from only one biological parent will be an unaffected carrier for the condition. Carriers function normally, usually exhibiting no symptoms of the disease, but they can pass the carrier gene on to their genetic offspring.
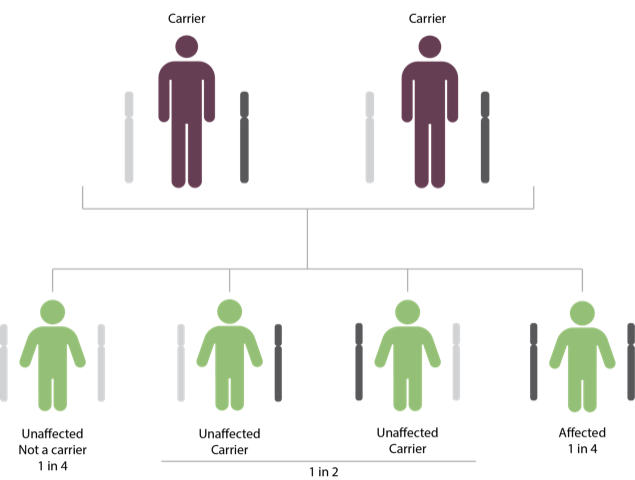
When both biological parents are carriers for the same recessive condition, each pregnancy has these risks (see diagram below):
- There is a 25% chance of inheriting the non-working gene from both biological parents and being affected with the disease.
- There is a 50% chance of inheriting one non-working gene and being an unaffected carrier.
- There is a 25% chance of inheriting no non-working genes (unaffected and non-carrier).
When only one biological parent is a carrier, the child has virtually no chance of being affected with the disease, but there is a 50% chance of being an unaffected carrier like the biological parent.
Genetic testing continues to evolve and donors screened at different times may have been tested with different screening panels. The details of each donor’s genetic testing is available for free through links on our donor catalog.
All donors are screened for Cystic Fibrosis carrier trait and for spinal muscular atrophy (SMA) carrier status. All donors are tested by Hemoglobin Electrophoresis to screen for Sickle-cell and Thalassemia carrier status and identify other hemoglobin-related blood disorders. Donors of Ashkenazi Jewish, French-Canadian, or Cajun descent also receive genetic carrier screening for Tay Sachs carrier trait. All Ashkenazi Jewish donors have been tested for a panel of conditions more common in their ethnic group.
Starting in June 2021, all new donors are tested using an Expanded Carrier Screening panel which includes all of the above conditions. This panel screens for over 260 different autosomal recessive conditions.
Donors screening through Expanded Carrier Screening may be positive as a carrier for one of the 260+ conditions tested. TSBC requires all recipients choosing a donor with positive carrier screening to complete an acknowledgement that they are aware of the test results. We strongly recommend recipients using a donor with one or more positive carrier results (1) receive genetic counseling and (2) consider carrier testing for themselves to reduce the risk of having an affected child.
There are a variety of labs that offer Expanded Carrier Screening. Each lab has different panels. If you wish to consider the lab we use for our Expanded Carrier Screening, you can learn more about Invitae’s screening options. We recommend you discuss testing options with a genetic counselor and/or your medical providers.
What if I know I am a carrier for a genetic condition?
Sometimes a recipient asks for additional screening for a donor after they have learned that they are a carrier for a condition through their own testing. We respond to registered recipient requests for additional donor carrier testing on a case-by-case basis. The decision can depend on a number of issues.
- Is an effective screening test available?
- Has the recipient received genetic counseling regarding her result?
- Is the donor available to be tested?
- Does the donor agree to be tested?
If additional screening can be performed for the donor, the recipient pays for any additional lab tests and all administrative fees for this service.
If you would like more specific information regarding testing for a specific donor, you may register as a recipient and once you have selected a donor, we can explore testing options for them.
You may also set up a genetic counseling appointment with our genetic counselor through the website. Specific information about your individual situation is best addressed in this way. Please read more information about these appointments, including how to schedule.
You may want to consider obtaining genetic counseling services for yourself through your own provider to ensure that you are aware of your own family history issues (if any), and to learn the risks, benefits and limitations of genetic testing for your pregnancy.
